Update – 27 October 2023
Background
Breast cancer
Breast cancer is the most common cancer in Australian women. In the year 2021, there were 19,886 new breast cancer diagnoses (accounting for more than one in four female cancer cases), and an estimated 3,138 women died from breast cancer.[1] Projections published by the Daffodil Centre in 2022 estimated that more than 90,000 Australian women will die from breast cancer between 2020 and 2044.[2] Breast cancer is relatively rare in males, with 164 cases diagnosed in 2021.[1]
Detecting and treating breast cancer at an early stage is key to saving lives and ensuring that women have the best possible quality of life after treatment. Given the burden of breast cancer in Australia, improvements in early detection could make a real difference to many people (the figure below).

At a population level, incremental improvements in early detection could significantly reduce the burden of breast cancer in Australia and make a real difference to many people.
BreastScreen Australia
The BreastScreen Australia program (BreastScreen) offers free two-yearly mammograms for asymptomatic women aged 50-74 years (available from age 40), aiming to detect breast cancers before they become symptomatic.
BreastScreen was introduced from 1991 and is a joint initiative of the Australian and state and territory governments. BreastScreen is jointly funded by the Australian Government and state and territory governments and delivered at a state and territory level by accredited service providers. The age-standardised participation rate remained between 53% and 54% from 2014–2015 to 2018–2019 and decreased due to the impact of the COVID-19 pandemic from March 2020 to 48% over 2020-2021.[3]
The primary purpose of BreastScreen is to reduce breast cancer mortality, and thanks to the nearly 2 million women who attend every two years, BreastScreen has led to a 21% reduction in population-level breast cancer mortality[4], with screened women having a 41–52% reduced risk of dying from breast cancer[5].
What is risk-based breast screening?
BreastScreen is targeted to females aged 50-74 on the basis that this population group will benefit most from regular mammographic screening. Within this age group, annual mammographic screening is offered to some participants based on risk factors such as their personal or family history of breast or ovarian cancer. Therefore, BreastScreen is already based on risks, to some degree.
However, with developments in both risk assessment and screening technologies, it is possible that BreastScreen could be more effective by providing more personalised screening protocols that are tailored according to risk (‘risk-based’ or ‘risk-stratified’ screening), aiming to find more breast cancers at an early stage before they spread or become symptomatic.
Many options are possible for identifying risk groups and then what might be offered to those risk groups. Important risk factors include high breast density (radiographically opaque breast tissue - see figure below), various genetic mutations (such as the BRCA1/2), hormonal and reproductive histories (e.g. age at first period, childbearing and breastfeeding history), alcohol consumption, some types of menopausal hormone therapy, and being overweight.
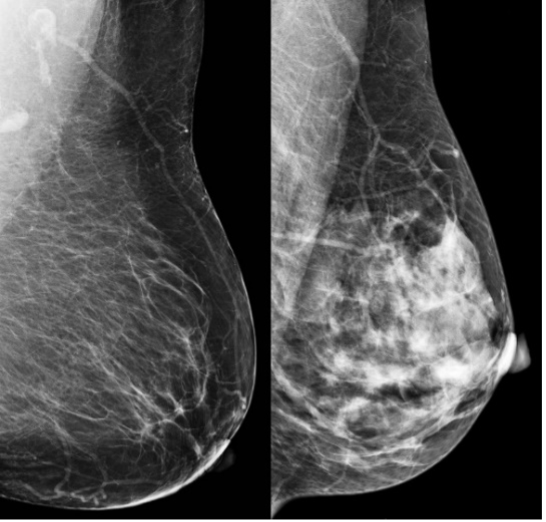
Mammograms (breast x-rays) showing low breast density (left) and high breast density (right)
Potential imaging tests include supplemental ultrasound, 3-D mammography, and imaging involving contrast (such as MRI tests and contrast-enhanced mammography). It is possible that risk-targeted use of additional or alternative breast imaging technologies could find more breast cancers earlier. It is also possible that different screening intervals could be offered, based on breast cancer risks.
Breast density is of particular interest because it is both a risk factor for breast cancer and higher breast density can also reduce the accuracy of mammograms. Some alternative breast imaging tests can be more accurate than mammography for women with dense breasts, so they are potential options for risk-based approaches to screening.
Changing a well-established and effective national program like BreastScreen Australia to offer risk-based screening protocols incorporating different risk assessments and screening tests would be a major undertaking. The evidence is complex and there are many considerations, including the potential benefits, harms and costs of different screening scenarios and what might be feasible and equitable in the Australian health and population setting.
References
- Australian Institute of Health and Welfare 2021. Cancer in Australia 2021. Cancer series no. 133. Cat. no. CAN 144. Canberra: AIHW. Accessed 09 March 2023.
- Luo Q, O'Connell DL, Yu XQ, Kahn C. et. al. Cancer incidence and mortality in Australia from 2020 to 2044 and an exploratory analysis of the potential effect of treatment delays during the COVID-19 pandemic: a statistical modelling study. Lancet Public Health. 2022 Jun;7(6):e537-e548.
- Australian Institute of Health and Welfare (2023) Cancer screening programs: quarterly data, AIHW, Australian Government, accessed 09 March 2023.
- Morrell S, Taylor R, Roder D, Dobson A. Mammography screening and breast cancer mortality in Australia: an aggregate cohort study. J Med Screen. 2012 Mar;19(1):26-34. doi: 10.1258/jms.2012.011127. Epub 2012 Feb 18. PMID: 22345322.
- Nickson C, Mason KE, English DR, Kavanagh AM. Mammographic screening and breast cancer mortality: a case-control study and meta-analysis. Cancer Epidemiol Biomarkers Prev. 2012;21(9):1479–88.
Project Personnel
Project Team
The ROSA Breast project was led by Associate Professor Carolyn Nickson, supported by a Project Coordination Group and a project team.
In 2018 the project established an Expert Management Group (EMG) of 30 members who helped establish the range of evidence to be collected, reviewed the interim findings, and identify priority activities for the next stage of work. The project later reduced this group to a smaller Expert Advisory Group (see more information below) of independent, multidisciplinary experts with ongoing consumer representation, retaining panels of co-opted experts to support specific activities. The project also gathered input from the BreastScreen Australia Program Management Group as well as a dedicated ROSA BreastScreen Reference Group comprising BreastScreen personnel from a range of work areas.
Project staff and their contributing roles are shown below (in alphabetical order). Staff were located at either the Daffodil Centre (a joint venture between Cancer Council NSW and the University of Sydney) or Cancer Council Australia.
Dr Denise Campbell, Systematic Reviewer
Prof Karen Canfell, Executive Lead
Ms Chelsea Carle, Systematic Reviewer and Policy Analyst
Dr Jennifer Cauchi, Research Coordinator
Dr Sabine Deij, Data Analyst and Health Economist
Mr Sam Egger, Statistician
Ms Victoria Freeman, Systematic Reviewer
Mr Paul Grogan, Policy and Strategy Advisor
Ms Suzanne Hughes, Systematic Reviewer
Dr Saima Islam, Data Analyst
Dr Qingwei Luo, Statistician
Dr Adelaide Morgan, Policy Analyst
A/Prof Carolyn Nickson, Project lead
Prof Dianne O’Connell, Statistician
Dr Lara Petelin, Data Analyst and Health Economist
Dr Pietro Procopio, Modeller
Elijah Tyeders, Research Assistant
Dr Andrea Smith, Implementation Scientist
Dr Julia Steinberg – Statistician and Epidemiologist
Ms Amanda Tattam, Communications and Stakeholder Engagement
A/Prof Natalie Taylor, Implementation Scientist
Ms Gabrielle Tiernan, Research Assistant
Paige Todd – Project Support
Dr Louiza Velentzis, Epidemiologist
Ms Emily Websdale, Project Support
Dr Susan Yuill, Systematic Reviewer
| Associate Professor Carolyn Nickson - Project lead | Lead, Breast Cancer Policy and Evaluation, the Daffodil Centre |
| Professor Karen Canfell – Executive Lead | Director, The Daffodil Centre |
| Professor Bruce Mann – Co-Chair - Expert Advisory Group | Professor of Surgery, The University of Melbourne Director of Breast Tumour Stream, Victoria Comprehensive Cancer Centre Director of Research, Breast Cancer Trials, Royal Melbourne & Women’s Hospital |
| Paul Vardon - Co-Chair Expert Advisory Group | Director, Cancer Screening Branch, Queensland Heath |
| Megan Varlow - Stakeholder Engagement, Strategy | Director, Cancer Control Policy, Cancer Council Australia |
| Paul Grogan - Project Adviser | Senior Strategic Adviser, The Daffodil Centre |
| Member | Affiliation/s |
| Prof Bruce Mann (Chair) | Professor of Surgery, University of Melbourne Director of Breast Tumour Stream, Victoria Comprehensive Cancer Centre |
| Mr Paul Vardon (Chair) | Director Cancer Screening Unit, Queensland Health Chair, BreastScreen Australia Program Management Group |
| Ms Alison Lang | Acting Director, Screening Policy Section, Cancer Policy and Services Branch, Australian Government Department of Health and Aged Care |
| Ms Harj Bariana | Director Clinical Services and BreastScreen Operations, Westmead Breast Cancer Institute, BreastScreen NSW Sydney West Service |
| Dr Jill Evans | Clinical Director and Chief Radiologist, Monash BreastScreen |
| Ms Anny Friis | Consumer Representative, Cancer Voices Australia (until June 2022) |
| Ms Leslie Gilham | Consumer Representative, Breast Cancer Network of Australia |
| Dr Mandy Henningham | First Nations Fellow, Cancer Council Australia and Postdoctoral Research Fellow in Indigenous Social Sciences, University of Sydney. |
| Ms Sarah McGill | Director – Screening and Prevention, Cancer Institute NSW |
| Associate Professor Vivienne Milch | Director Cancer Care, Cancer Australia Medical Advisor to the Department of Health and Aged Care on cancer screening policy |
| Associate Professor Michelle Reintals | Head of RANZCR Breast Imaging Group, Clinical Director, BreastScreen South Australia. |
| Prof David Roder | Chair, Cancer Epidemiology and Population Health, School of Health Sciences, University of South Australia Principal Research Fellow, SA Health and Medical Research Institute |
| Prof Christobel Saunders | James Stewart Chair of Surgery Royal Melbourne Hospital University Department of Surgery | School of Medicine |
Further Information
Publications
In addition to the various detailed technical reports provided to the Australian Government, the ROSA project has led or contributed to the recent peer-reviewed publications below:
- Velentzis LS, Freeman V, Campbell D, Hughes S, Luo Q, Steinberg J, Egger S, Mann GB, Nickson C. Breast Cancer Risk Assessment Tools for Stratifying Women into Risk Groups: A Systematic Review. Cancers (Basel). 2023 Feb 9;15(4):1124.
- Bulliard JL, Beau AB, Njor S, Wu WY, Procopio P, Nickson C, Lynge E. Breast cancer screening and overdiagnosis. Int J Cancer. 2021 Apr 19. Epub ahead of print. PMID: 33872390.
- Feletto E, Grogan P, Nickson C, Smith M, Canfell K. How has COVID-19 impacted cancer screening? Adaptation of services and the future outlook in Australia. Public Health Res Pract. 2020 Dec 9;30(4):3042026.
- Saxby K, Nickson C, Mann GB, Park A, Bromley H, Velentzis L, Procopio P, Canfell K, Petrie D. Moving beyond the stage: how characteristics at diagnosis dictate treatment and treatment-related quality of life year losses for women with early stage invasive breast cancer. Expert Rev Pharmacoecon Outcomes Res. 2021 Aug;21(4):847-857. Epub 2021 Jan 27.
- Saxby K, Nickson C, Mann GB, Velentzis L, Bromley HL, Procopio P, Canfell K, Petrie D. The financial impact of a breast cancer detected within and outside of screening: lessons from the Australian Lifepool cohort. Aust N Z J Public Health. 2020 Jun;44(3):219-226. Epub 2020 Apr 20.
- Lew JB, Feletto E, Wade S, Caruana M, Kang YJ, Nickson C, Simms KT, Procopio P, Taylor N, Worthington J, Smith D, Canfell K. Benefits, harms and cost-effectiveness of cancer screening in Australia: an overview of modelling estimates. Public Health Res Pract. 2019 Jul 31;29(2):29121913.
- Nickson C, Velentzis LS, Brennan P, Mann GB, Houssami N. Improving breast cancer screening in Australia: a public health perspective. Public Health Res Pract. 2019 Jul 31;29(2):2921911
Mailing list
To receive updates regarding the project, please contact rosabreast@nswcc.org.au.

