Cancer research is an important part of cancer care and over the years has let to better treatments and outcomes for people diagnosed with cancer. A clinical trial is one type of cancer research and helps to show if a new approach to prevention, screening, diagnosis or treatment works better than current methods.
What is a clinical trial?
The World Health Organization defines a clinical trial as "any research study that prospectively assigns human participants or groups of humans to one or more health-related interventions to evaluate the effects on health outcomes." Put simply, a clinical trial is a study that compares responses to different interventions in real settings, to test the effectiveness of medicines or other health measures. Clinical trials are voluntary and are governed by strict rules and ethics.
They are often undertaken in a clinical setting, such as a hospital or outpatient clinic. The participants are usually patients, but they may include former patients and people who are well.
Clinical trials are an important way to improve treatment for people with cancer - and the only way to thoroughly evaluate the effects of a clinical intervention. If a clinical trial proves that a test or treatment is more effective than existing options, it may become the new standard of care for patients in the future. A trial can also identify potential risks and side effects.
Who can participate in a clinical trial?
There are guidelines for each clinical trial about who can join, called eligibility criteria. Clinical trials are open to adults and children, however children under 18 need the permission of their parents or guardians to be involved.
Most cancer research involves current patients. However, some studies focus on cancer survivors, carers, family members, people at risk of cancer or people who have not been affected by the disease.
To ensure that results reflect Australia’s diverse population, it is important that research involves people of all ages, genders and sexualities, as well as people from a wide range of economic, social, racial and cultural backgrounds.
Trials can be conducted over a number of years, and it can take some time for the results to be published in medical journals and other science literature.
Participation is voluntary, and you can withdraw from a study at any time. If you pull out, you will still be able to receive the type of treatment that is currently the best option for you.
Where does research take place?
Cancer research is carried out in many places including hospitals, treatment centres, universities and laboratories.
You can sometimes be involved in cancer research from home. For instance, you may have treatment or medicine mailed to you, or you may be asked to fill in a survey or complete a face-to-face or telephone interview. In Australia, some people are now taking part in clinical trials via telehealth.
What do I need to know before beginning a clinical trial?
Researchers must provide written information about the clinical trial to anyone who is thinking about participating, including:
- the purpose and phase of the trial
- who is running the study
- who has approved the research
- who is funding the study
- how the study will be run
- what will be required of a participant (including length of involvement and potential side effects or other risks)
- information about your rights, such as confirming that medical records are private and confidential
- contact details for the person who will be able to explain who may have access to your information and for what purpose.
It is a legal requirement that all personal information collected as part of a clinical trial is stored securely for at least 15 years before it can be destroyed.
If you decide to participate, you will first have to give your informed consent by signing a form, confirming that you have read and understood the purpose, risk and possible outcomes of the trial.
Are clinical trials safe?
All clinical trials in Australia are regulated by an international standard of practice, called Good Clinical Practice. These standards ensure trials are conducted to a high scientific standard and that they are safe for people taking part.
Several committees examine and approve a clinical trial before it commences, and confirm that the study is considered to be of scientific merit and fair (ethical).
Researchers must follow strict guidelines to make sure studies are as safe as possible for everyone involved - this is called their duty of care.
What is a randomised controlled trial?
It is important for researchers to know that the results of a study are accurate and not caused by chance.
Many clinical trials are randomised control trials (RCTs). A randomised controlled trial helps prevent bias making it the best way to test if a new treatment works. Randomised controlled trials are designed to eliminate biases by "randomly" selecting participants and "controlling" for other factors.
Typically, participants in a clinical trial will be put in one of two groups:
- test or experimental group - are given the new treatment being assessed;
- control group (or arm) - are given an established treatment that is already in use, other standard care or a placebo.
Will I be given a placebo?
Placebos (inactive or fake treatment) are made to look, taste or feel like the treatment being tested. Examples include sugar pills and saline solutions.
Placebos are used to show if any improvements are because of the actual treatment or because of other factors such as being more closely monitored or expecting the treatment to be helpful.
You will be told if a study uses a placebo but you will not be told which treatment you are having.
What is a blind study?
When participants don't know which study group they are in, this is called a blind study. Some randomised controlled trials are called double-blind studies - this is when both the trial team and participant don't know which group the participant is in.
If your doctor needs to find out what treatment you're having for safety reasons they are able to find out by contacting the trial organisers.
What does it cost to participate?
Participation in a clinical trial is free for Australian residents and citizens. There may be out-of-pocket expenses, such as travel, which may also be reimbursed to you. You will still need to pay for any treatments or tests you would normally pay as part of your standard care.
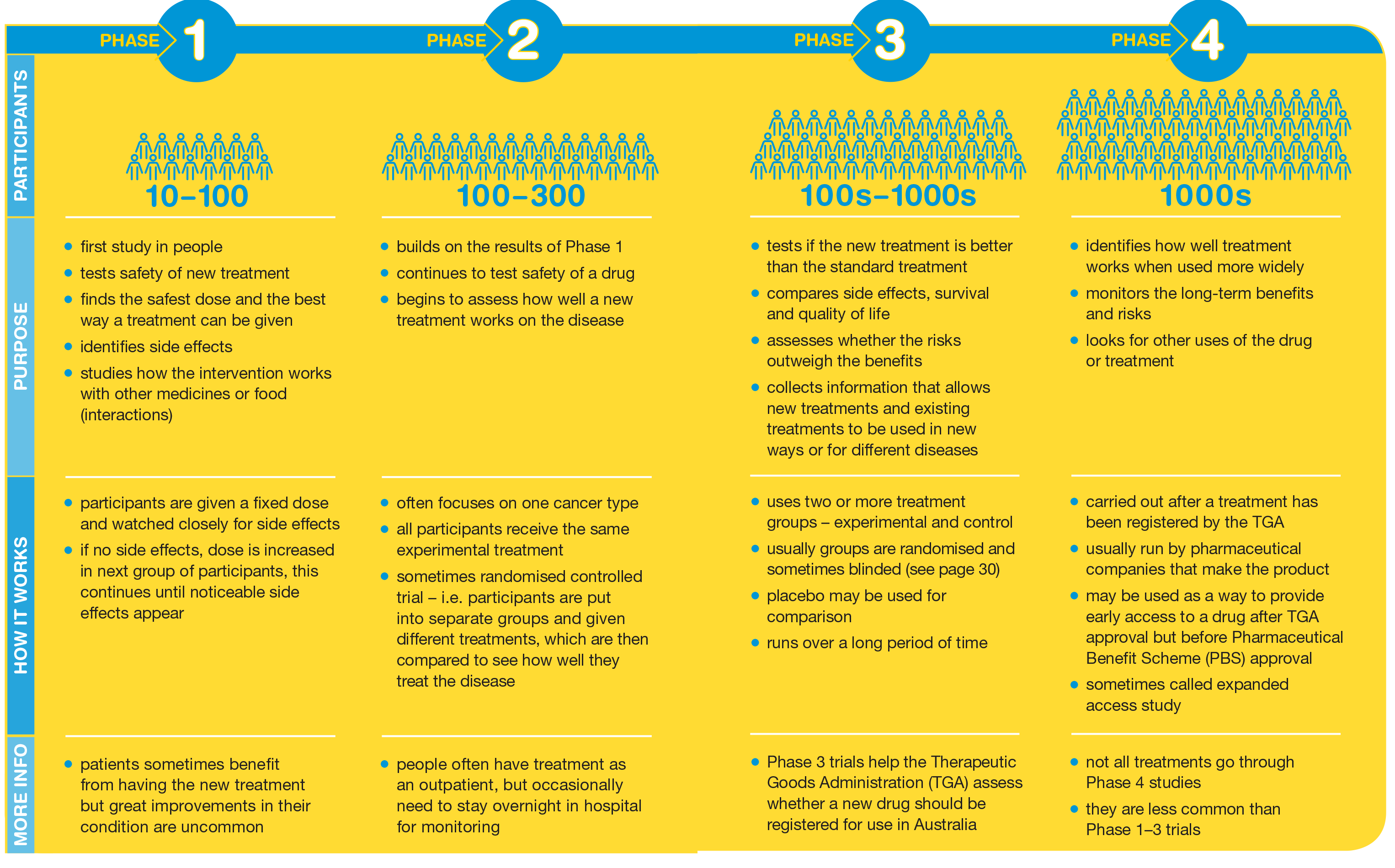
What do the different clinical trial "phases" mean?
Researchers may spend years developing new treatments and medicines before involving people. Clinical trials are then developed to progress in a series of steps called "phases". There are generally four phases and the information gathered in each determines if the study can move on to the next phase. See the illustration below for more information on what happens during a clinical trial.
Clinical trials team
If you join a study, you may have contact with some of the following professionals:
- medical specialists such as oncologists, surgeons and haematologists
- clinical trials nurse or research nurse
- clinical trials or study coordinator
- contact person for you during the trial
- researcher or investigator
- principal investigator
- allied health practitioners such as pharmacists, counsellors or dietitians.
You should also be given details of the approving Ethics Committee and trial secretariat.
Support during and after the trial
All studies include a contact person, who is often the clinical trials nurse or study coordinator. However, you will also be given access to a person independent of the research team, in case you have a complaint about the study. This is one of the participant's rights of joining a study.
After the trial is finished, researchers may stay in contact with you for some time so they can collect long-term information on your health.
Finding a study
There are many ways to find out about a clinical trial. Most cancer specialists will know about current studies and may recommend you join a suitable study. You can also ask them if there are any potential trials you could consider.
The Australian New Zealand Clinical Trials Registry (ANZCTR), is an online register of clinical trials being undertaken in Australia, New Zealand and elsewhere. It includes trials from the full spectrum of therapeutic areas of pharmaceuticals, surgical procedures, preventive measures, lifestyle, devices, treatment and rehabilitation strategies, and complementary therapies.
The government cancer agency, Cancer Australia, also has online information about current cancer clinical trials, in partnership with the Australian New Zealand Clinical Trials Registry, the University of Sydney and consumer group Cancer Voices.
Questions to ask
Even though it is mandatory for a clinical trial to supply patient information about the trial, it is important that you ask any questions you may have before joining a study. Examples of questions include:
- What is being tested in the trial and why?
- Who will oversee my cancer care while I am participating?
- Where will I receive treatment and for how long?
- Who do I contact if there is a problem?
- What are my chances of benefitting from this research?
- What are the risks to me?
- Will I experience any side effects? How will they be treated?
- What type of tests are involved?
- How long does the study go for?
- Can I receive reimbursement for out-of-pocket expenses?
- If I join this study, will I miss out on other treatment opportunities later?
- Can I still take other medication or complementary therapies while I am involved in the trial?
- Who will have access to my personal information as part of the trial?
- How much time do I have to decide if I want to participate? Where can I get reliable information?

